产品说明
一般描述
Pharmaceutical secondary standards for application in quality control provide pharma laboratories and manufacturers with a convenient and cost-effective alternative to the preparation of in-house working standards
分析说明
These secondary standards offer multi-traceability to the USP, EP and BP primary standards, where they are available.
其他说明
This Certified Reference Material (CRM) is produced and certified in accordance with ISO 17034 and ISO/IEC 17025. All information regarding the use of this CRM can be found on the certificate of analysis.
基本信息
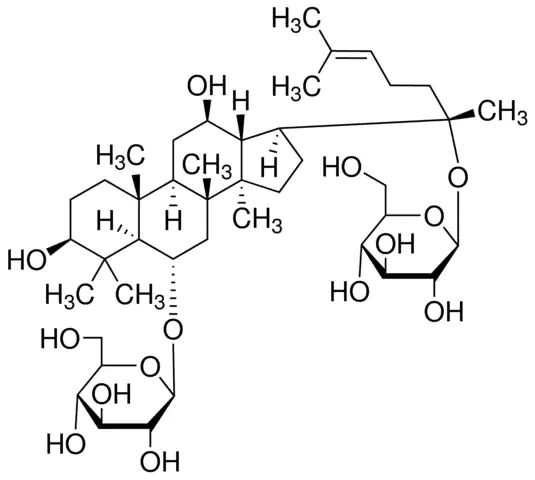
| 经验(实验)分子式 | C42H72O14 |
| 分子量 | 801.01 |
| MDL编号 | MFCD00210293 |
产品性质
| 等级 | certified reference material pharmaceutical secondary standard |
| 描述 | Pharmaceutical Secondary Standard; Certified Reference Material |
| 包装 | pkg of 50 mg |
| 格式 | neat |
| 药典可追溯性 | traceable to USP 1291672 |
| 储存温度 | 2-8℃ |
| SMILES string | C\C(C)=C/CC[C@](C)(O[C@@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O)[C@H]2CC[C@]3(C)[C@@H]2[C@H](O)C[C@@H]4[C@@]5(C)CC[C@H](O)C(C)(C)[C@@H]5[C@H](C[C@@]34C)O[C@@H]6O[C@H](CO)[C@@H](O)[C@H](O)[C@H]6O |
| InChI | 1S/C42H72O14/c1-20(2)10-9-13-42(8,56-37-34(52)32(50)30(48)25(19-44)55-37)21-11-15-40(6)28(21)22(45)16-26-39(5)14-12-27(46)38(3,4)35(39)23(17-41(26,40)7)53-36-33(51)31(49)29(47)24(18-43)54-36/h10,21-37,43-52H,9,11-19H2,1-8H3/t21-,22+,23-,24+,25+,26+,27-,28-,29+,30+,31-,32-,33+,34+,35-,36+,37-,39+,40+,41+,42-/m0/s1 |
| InChI key | YURJSTAIMNSZAE-HHNZYBFYSA-N |
安全信息
| 象形图 |  |
| 警示用语: | Warning |
| 危险声明 | H302 |
| 预防措施声明 | P264 - P270 - P301 + P312 + P330 - P501 |
| 危险分类 | Acute Tox. 4 Oral |
| 储存分类代码 | 13 - Non Combustible Solids |
| WGK | WGK 3 |
| 闪点(F) | Not applicable |
| 闪点(C) | Not applicable |